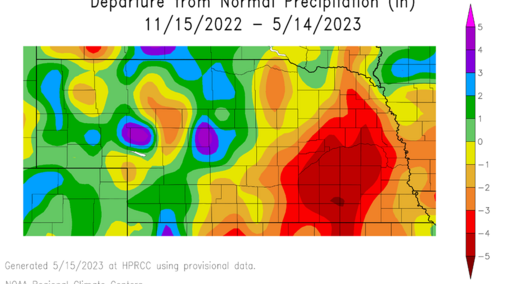
There have been some reports of corn seedling burn with anhydrous ammonia application in the state. This injury is more likely in areas with limited rainfall after fall or spring anhydrous application, as some parts of the state have received less rainfall than others (Figure 1). Here we suggest some practices to minimize crop injuries.
Causes of Crop Injury
The crop injuries are of three types: ammonia gas injury from applying anhydrous ammonia, very high soil pH following anhydrous ammonia application, and salt damage (Figure 2). Ammonia gas injury is a concern when the germinating or emerging crop is in place during anhydrous ammonia application. Germinating seeds may be damaged or even killed when the band is within four to six inches of the row, depending on the soil sand content and the application rate. If the injection band is shallow and not well covered, especially with dry soil, sufficient ammonia escape can occur to damage plants. Plants may also be damaged if ammonia flow occurs when the applicator is raised from the soil such as at row ends.

Very high and potentially damaging soil pH occurs following anhydrous ammonia application. The ammonia (NH3) quickly reacts with soil water (H2O) to produce ammonium (NH4+). The NH4+ reacts with the negative charge of soil to minimize loss to volatilization and to have low leaching potential. However, the reaction of NH3 with H2O to produce NH4+ results in the release of hydroxyl ions (OH-) and a great increase in soil pH around the anhydrous band. The high soil pH can be damaging to nearby germinating seeds and roots.
The salt effect resulting from band application of fertilizer-N and other nutrients may be of greatest concern. This is addressed in more detail in NebGuide G361 on starter fertilizers. Band application of fertilizer-N results in high concentration of NH4+ plus nitrate (NO3-) with NO3- increasing with nitrification of NH4+. The NH4+ and NO3- react with other ions in soil solution to raise the salt concentration, commonly measured as electrical conductivity (EC). The high salt concentration can be damaging if the fertilizer band is too near the seed and roots of growing plants. The high salt effect is relatively more persistent while the risk of ammonia gas injury is very short and the high pH effect is of intermediate duration.

Crop Symptoms
Typically, roots experiencing excessive concentrations of salts or ammonia will have root tips which are blunted and brown or black (Figure 2). The plants will often be stunted and may have leaves that are purple or reddish-tinted (Figure 3).
How to Minimize the Injury
The salts could be dispersed with rainfall and irrigation. If plants continue to show symptoms of salt damage, irrigate with an inch or two of water if possible. However, the best solution is to avoid creating the potential for damage in the future by keeping fertilizer-N injection bands, other than low-rate starter bands, several inches away from the seedling.
Note: This article is a revised version of previously published article in 2020.